15-05
A.A.C. Bode, P.G.M. Pulles, M. Lutz, W.J.M. Poulisse,
S. Jiang, J.A.M. Meijer,
W.J.P. van Enckevort, E. Vlieg,
Sodium chloride dihydrate crystals: morphology, nucleation, growth, and
inhibition,
Cryst. Growth Des. 15 (2015) 3166-3174
Abstract:
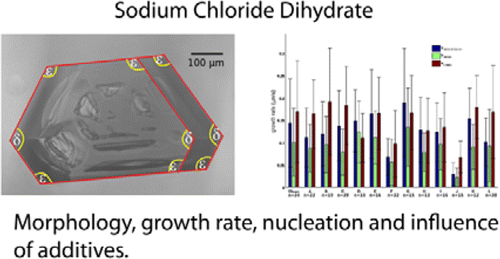
Sodium chloride powders have
a strong tendency to cake at freezing temperatures, which is caused by the
formation of sodium chloride dihydrate solid bridges
between the powder particles. Using the crystallographic structure, the
morphology of the dihydrate crystals was determined.
In addition, the growth rate of the most important facets of these crystals was
measured as well as the influence of a number of anticaking agents and related
additives on the growth rate. A likely candidate for the inhibition of dihydrate growth was found: a mixture of iron(III) and
l-tartaric acid, which probably forms a metal–organic coordination complex. The
nucleation rate of the dihydrate was found to be
extremely low and to increase with time. This is probably caused by the formation
of a metastable prenucleation phase, which inhibits
the nucleation of dihydrate crystals. The influence
of the additives on the nucleation rate was found to be limited.