16-08
W.W. Li, L. Spix, S.C.A. de
Reus, H. Meekes, H.J.M. Kramer, E.
Vlieg, J.H. ter Horst,
Deracemization of a racemic compound via Its
conglomerate-forming salt using temperature cycling,
Cryst. Growth Des. 16 (2016) 5563-5570
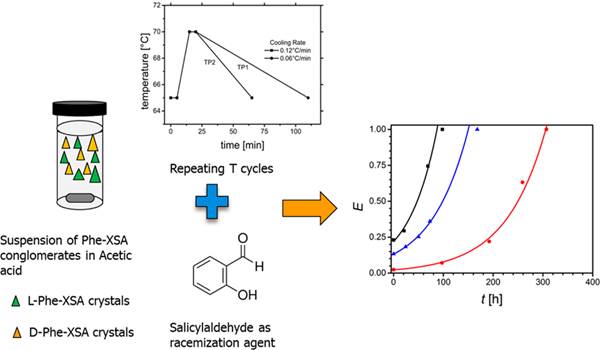
Abstract:
Salts of chiral molecules, which
originally crystallize as racemic compounds, could form conglomerates. The
utilization of such conglomerate salts, as
intermediates for the deracemization of corresponding
racemic compounds, expands the theoretical application range of Viedma Ripening by roughly 10-fold. In the present study,
the use of temperature cycling on conglomerate forming salts as an alternative
technique for Viedma Ripening was studied. The
racemic compound Phenylalanine (Phe) was successfully
deracemized via its conglomerateforming
salt with 2,5-xylenesulfonic acid (XSA) by continuous
heating−cooling cycles applied to its suspension
in glacial acetic acid, coupled with a solution racemization reaction. In
addition, the dependence of the deracemization rate
on the operational parameters was studied. Enhanced racemization reaction
kinetics, either by a larger amount of free amino acid or by a higher
concentration of catalyst, was shown to accelerate the deracemization
process. It seems to indicate that a concentration difference between the two
enantiomers, which could be diminished by a faster racemization rate, behaves as
one of the major rate-limiting factors for the deracemization
process. A larger mass fraction of solid dissolving and recrystallizing in the
heating cooling cycles, achieved by either a larger temperature swing or a
smaller dry mass concentration, also leads to a faster deracemization.
A change in cooling rate does not affect the deracemization
rate significantly within the range tested, indicating a limited presence of
secondary nucleation of the minor enantiomers. The results can be used as a
preliminary foundation for process
optimization as well as mechanisms investigation. The advantages and
disadvantages of temperature cycling and Viedma
Ripening, as deracemization methods in an industrial
setting, are discussed.