A.H.J.
Engwerda, N. Koning, P. Tinnemans, H. Meekes, F.M. Bickelhaupt, F.P.J.T.
Rutjes, E. Vlieg,
Deracemization of a racemic allylic sulfoxide using Viedma ripening,
Cryst. Growth Des. 17 (2017) 4454-4457
Abstract
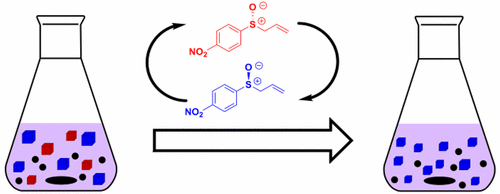
Despite the importance of enantiopure chiral
sulfoxides, few methods exist that allow for their deracemization. Here, we
show that an enantiopure sulfoxide can be produced from the corresponding
racemate using Viedma ripening involving rearrangement-induced racemization.
The suitable candidate for Viedma ripening was identified from a library of 24
chiral sulfoxides through X-ray structure determination. Starting from the
racemic sulfoxide, an unprecedented application of a 2,3-sigmatropic
rearrangement type racemization in a Viedma ripening process allowed for
complete deracemization.