O.I. van den
Boomen, R.G.E. Coumans, N. Akeroyd, T.P.J. Peters, P.P.J. Schlebos, J. Smits,
R. de Gelder, J.A.A.W. Elemans, R.J.M. Nolte, A.E. Rowan,
Carbenoid transfer reactions catalyzed by a ruthenium porphyrin Macrocycle,
Tetrahedon 73 (2017) 5029-5037
Abstract
A
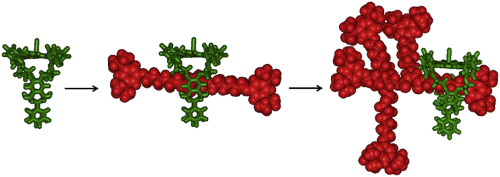
ruthenium porphyrin functionalized with a cavity
based on diphenylglycoluril is applied as a catalyst in carbenoid transfer
reactions using α-diazoesters as substrates. The latter compounds contain
a blocking group connected via an α,ω-dioxyalkyl spacer of 3 or 6
carbon atoms. The reaction of an excess of the α-diazoester with the short
spacer with the ruthenium porphyrin macrocycle leads to two products, a
[2]rotaxane and a maleate ester, which are the result of dimerization reactions
at the inside and the outside of the cavity, respectively. A similar reaction
using the α-diazoester with the long spacer also yields high molecular
weight species. Mass spectrometric and NMR studies suggest that C![]() H and/or C
H and/or C![]() C insertion reactions take place on the thread of
the initially formed rotaxane. It is proposed that these reactions are favoured
by effective molarity effects because of the close proximity of reactive
species in the interlocked geometry.
C insertion reactions take place on the thread of
the initially formed rotaxane. It is proposed that these reactions are favoured
by effective molarity effects because of the close proximity of reactive
species in the interlocked geometry.